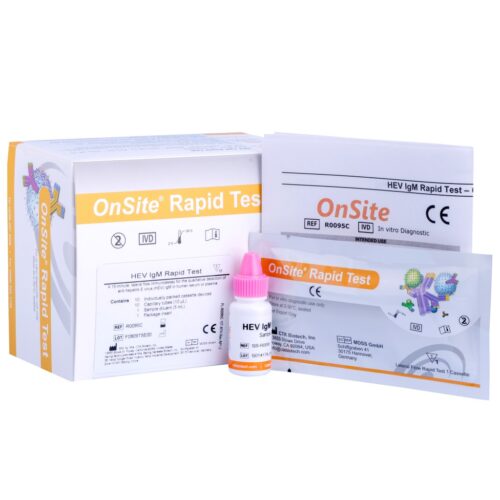
RecombiLISA HEV IgM ELISA kit is for qualitative detection of anti-hepatitis E virus IgM in human serum or plasma.
Features of HEV IgM ELISA Kit:
- Utilizes IgM capture technique.
- Suitable for diagnosing acute HEV infection.
- Assay time: 75 Mins.
- Sensitivity: 100% & Specificity: 100%.
- Utilizes IgM capture technique.
- Suitable for diagnosing acute HEV infection.
- Assay time: 75 Mins.
- Sensitivity: 100% & Specificity: 100%.
Intended use of HEV IgM ELISA Kit:
The RecombiLISA HEV IgM ELISA is a solid-phase enzyme-linked immunosorbent assay for the qualitative detection of anti-hepatitis E virus (HEV) IgM in human serum or plasma. It is intended for professional use only and to be used as an aid in the diagnosis of infection with HEV.
Introduction of HEV IgM ELISA Kit:
Hepatitis E, a major form of enterically transmitted hepatitis, is widespread in many developing countries but is currently considered an emerging threat to other parts of the world. HEV is a non-enveloped, positive-sense, single-stranded RNA virus. It is currently classified within the family Caliciviridae. It is mainly transmitted through the fecal-oral route. At least four major genotypes of HEV have been recognized: Genotypes 1 and 2 are restricted to humans while genotypes 3 and 4 can infect both humans and animals. Antibody response peaks approximately one month after initial infection. Antiviral IgM is detected in >90% patient and persists for 3 months. Anti-HEV IgM is also a well-established marker of recent infection and is the most convenient one for diagnosis.
RecombiLISA HEV IgM ELISA Kit:
The HEV IgM ELISA Kit is an enzyme-linked immunosorbent assay (ELISA) designed for the qualitative detection of IgM-class antibodies to the hepatitis E virus (HEV) in human serum or plasma.
Purpose: The HEV IgM kit is used in clinical laboratories for the diagnosis and management of patients related to HEV infection.
Virus Background:
- Hepatitis E virus (HEV) is a non-enveloped, single-stranded RNA virus.
- HEV infections can lead to acute or sub-clinical liver diseases similar to hepatitis A.
- Endemic in developing countries, HEV is also seen sporadically in developed countries, sometimes without a history of travel to endemic areas.
- Pregnant women are at higher risk, with case-fatality rates ranging from 0.5% to 3% and even higher (15% to 25%) among pregnant individuals.
Principle of the Assay:
- The kit follows a two-step incubation, solid-phase antibody capture ELISA approach.
- Polystyrene microwell strips are pre-coated with antibodies directed to human immunoglobulin M proteins (anti-μ chain).
- During the first incubation, any IgM-class antibodies present in the patient’s serum or plasma are captured in the wells.
- After washing out other components (including IgG-class antibodies), specific HEV IgM antibodies captured on the solid phase are detected.
- Recombinant HEV ORF2 antigens conjugated to horseradish peroxidase (HRP-Conjugate) are used for detection.
- Chromogen solutions are added, resulting in a color change that is proportional to the amount of antibody captured in the wells.
Clinical Application:
- The HEV IgM kit is particularly useful for diagnosing the acute phase of HEV infection, where IgM antibodies are generated before other immunoglobulins.
- It can also be used for follow-up of HEV-infected patients.
Storage and Expiry:







Reviews
There are no reviews yet